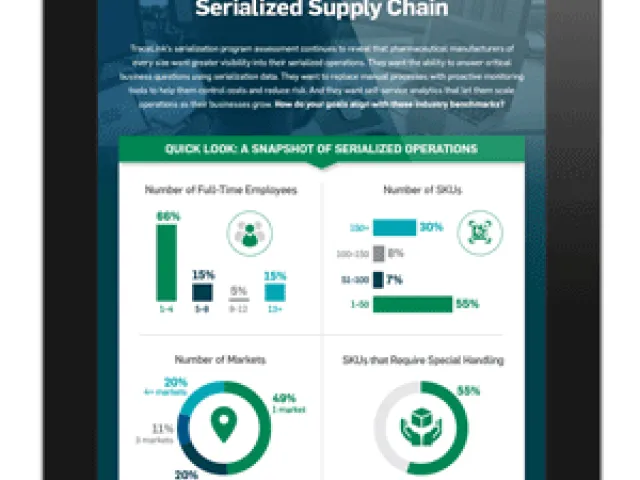
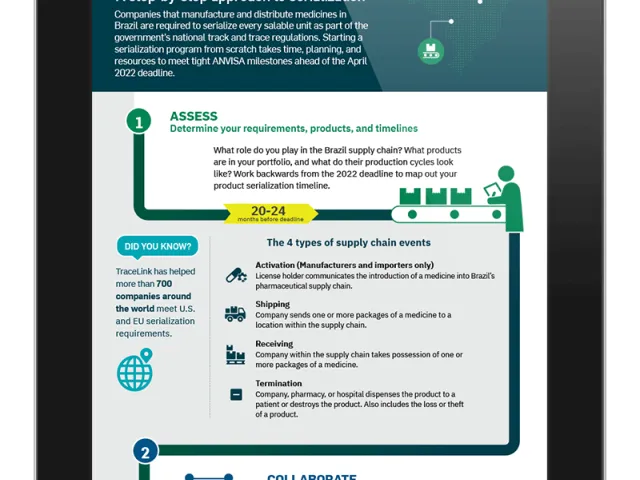
Table of contents
As product traceability and reporting requirements in China continue to be finalized—and as new compliance deadlines are announced—companies that manufacture and distribute products for the China market must balance a “wait-and-see” approach with the need to move quickly to secure or maintain their market presence. One decision that companies can—and should—be weighing immediately is how they will implement an effective, long-term product serialization strategy.
In packaging products for sale in China, companies should consider several important factors:
China allows two different serialization and encoding standards
China’s original law, published in 2012 and phased in by 2015, defined a single Electronic Drug Monitoring Code (EDMC), based on the Chinese Drug Electronic Administration (DEA/eCode) standard: a 20-character encoding standard that includes a 7-character drug identification code. Following a series of industry challenges, China’s National Medical Products Administration (NMPA) issued a revised and expanded set of technical guidelines in 2019 that included updated serialization and encoding standards, renamed as the Drug Traceability Code (DTC).
The new guidelines included a second DTC option that conforms to ISO/IEC 15459 specifications—the same specifications as the GS1 coding standard used by manufacturers to meet traceability regulations in markets around the world. In effect, companies that follow GS1 encoding standards will be able to comply with the China product serialization and identification requirements.
There are multiple ways you can apply the product identifier
Companies selling medicines in China must also consider the cost and logistics of applying the drug traceability code to the physical unit-level packaging—as well as to cases, cartons, and pallets of aggregated products. While the Chinese DEA/eCode standard uses a linear 1-dimensional barcode format, companies that decide to apply the ISO/IEC format can choose between several types of data carriers: a 1-dimensional barcode, a 2-dimensional “data matrix” barcode, or a radio-frequency identifier (RFID) tag. NMPA regulations also require that the carrier include human-readable information.
Some product data elements are required, some are optional
The next decision involves exactly what product data you will include in the product identifier. NMPA regulations require two data elements: a Drug Identification Code and a unique serial number. Once you determine your packaging configuration for China—including saleable units, packs, and cases—this data is part of the information that must be shared with NMPA as part of determining unique drug ID codes for the products and packages in your product portfolio. And, while the unit’s serial number is a required element, NMPA guidelines also allow additional optional elements that include lot number, manufacturing date, and expiry date.
Supply chain readiness in China should determine your serialization strategy
Decisions about product identifiers—format, carrier, and data—depend on the ability of your end-to-end supply chain—including CMOs, CPOs, 3PLs, distributors, and dispensers—to process serialized and aggregated products. As noted, China’s initial law that took effect in 2015 spurred the adoption of the original single Electronic Drug Monitoring Code (EDMC) across the China supply chain. With the revised 2019 law, the China supply chain faces another period of transition as it adapts to expanded product traceability and reporting requirements—and the introduction of a second DTC option that is GS1-compatible.
Companies that are currently selling products in China—or that plan to enter the China market—need to assess their distribution network to determine if and how their product packaging will meet “on-the-ground” business requirements. Key questions include:
- Are your distributors’ operations limited to DEA/eCode identifiers? Do they plan to add ISO/EIC capabilities—and when?
- Given current China supply chain considerations, should you consider a transition strategy that starts with eCoded products today but that can change as the market adopts of GS1 standard.
- Is there a viable distribution network for your China products that can handle ISO/EIC encoding so you don’t have to reconfigure existing GS1-based packaging lines?
- Is it cost-effective to maintain dedicated DEA/eCode packaging lines for products that are specific to the China market?
- If you plan to transition your current China serialization strategy to take advantage of GS1-compatible encoding, what kind of education programs can you provide to help prepare supply chain partners for the transition?
- Do distributors or dispensers want lot numbers and expiration dates to streamline warehouse or inventory operations?
Customize your China strategy with TraceLink
Since 2013, TraceLink has been helping companies serialize products and meet traceability compliance regulations for the China market. TraceLink’s regulatory expertise and proven serialization solutions give you the flexibility you need to meet China’s complex legal and regulatory requirements and adapt to fast-changing business conditions. Rely on TraceLink to stay up-to-date on the latest China technical requirements and compliance deadlines—and for insight into how the industry is balancing the challenges and opportunities in this dynamic market.