Table of contents
On July 13, Brazil’s Health Regulatory Agency, ANVISA, released the latest draft of its Normative Instruction for meeting the country’s drug traceability and reporting requirements. While submitting a Serialization Plan will still be required, the draft as released dispenses with any interim measures described in previous versions—including the “implementation phase”— ahead of the April 2022 deadline as well as phased-in requirements based on priorities or classification of serialized products. Key provisions include:
- The Normative Instruction applies to all members of the drug supply chain: Manufacturers, Distributors, and Dispensers (with few exceptions). (Article 2)
- Registry holders (Marketing Authorization Holders, or MAHs) must enter and maintain product master data, commercialization information, and metadata such as Anatomical Therapeutic Chemical (ATC) codes in the National Medicine Control System (SNCM) portal. (Article 3)
- The drug exemption list remains unchanged from the previous version of the Normative Instruction. (Article 4)
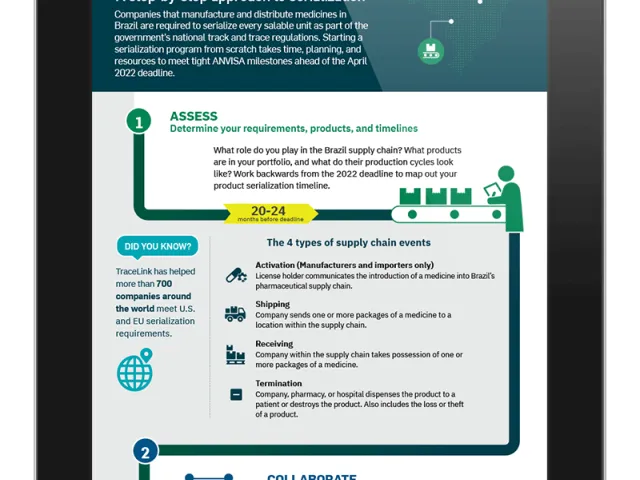
- By April 28, 2022, all non-exempt drugs must be serialized in order to record supply chain movements to the SNCM. (Article 4)
- Manufacturers and importers must create and maintain a serialization plan in the SNCM portal for in-scope production lines and medicines. They must also account for “partial percentages” of serialized production lines until complete serialization is achieved by April 28, 2022. (Article 5)
- Starting April 28, 2022, all supply chain members must report events on all serialized and commercialized drugs to the SNCM. (Article 6)
- Supply chain stakeholders will need to adapt to the technical specifications for reporting to the SNCM and that the SNCM operator may adopt complementary solutions as the system develops. (Article 7)
April deadline, short timeline have companies assessing fastest course
The final draft mirrors the law as far as requirements, scope, and deadlines are concerned, with no additional exemptions or allowances based on industry readiness. With the April 2022 deadline firmly established by ANVISA, companies should no longer delay their preparation—and risk non-compliance—based on the possibility of a legislative change to the legal deadline and the time it would take to enact such a change.
An immediate start and a focus on core serialization and reporting capabilities should enable companies to meet ANVISA requirements. However, they should also be planning for ongoing modifications to the SNCM system, per Article 7, as well as the need for trade partner data exchange—horizontal integration—to ensure accurate reporting and business continuity across a fully serialized supply chain.
Brazil compliance: What companies can do now
What can TraceLink customers do now to prepare for Brazil’s traceability requirements?
- Register their medicines with ANVISA and begin uploading their product and company master data required for SNCM reporting.
- Start their implementation projects with TraceLink to map use cases and identify workflow triggers for Activation and Shipment reports.
- Work with TraceLink’s Network Success Team to onboard and connect their trading partners.
TraceLink customers also have access to bi-weekly Brazil Special Interest Group meetings where they can discuss use cases and reporting workflows; get regular updates on TraceLink’s Brazil compliance solution; and learn how TraceLink’s OPUS platform will support faster deployment of new software releases as the SNCM system evolves. Beginning in September, the SIG will transition to a Brazil Innovation Forum where customers can share ideas for product feature enhancements with TraceLink product and service managers.
Using a 3PL in Brazil? Factor in additional time and complexity.
Companies using a third-party logistics provider (3PL) in Brazil face even greater challenges in ensuring smooth operations by the April deadline. TraceLink 3PL integration projects should be started immediately: making sure your 3PL is up to speed on the requirements; supporting secure partner on-boarding and connectivity; and developing 3PL transaction software design and implementation. To ensure that your 3PL will be able to support their customers, a regular review of 3PL readiness is critical to staying on schedule and mitigating risk.
Rely on TraceLink for proven solutions and support
TraceLink’s Brazil solution builds on our proven core compliance application with workflows and reporting tools that can be easily configured to enable multinational and Brazilian domestic pharmaceutical manufacturers—and agents or 3PLs that support them—to meet ANVISA requirements for product serialization and SNCM reporting.
TraceLink’s Serialized Traceability solution lets companies exchange product data with trade partners to streamline the movement of serialized and aggregated products and minimize delays due to SNCM reporting errors. And TraceLink’s network-tenant solution architecture means your Brazil solution is continuously updated to support changing regulatory and business requirements. Contact TraceLink to join the TraceLink Brazil Special Interest Group and learn more about our Brazil compliance solutions.