Table of contents
Key Takeaways
- More than 43% of pharma supply chain companies suffer from a lack of supply chain agility.
- Actionable visibility and contextually accurate agility will make your supply chain more resilient to disruptions.
- A single platform for collaboration. data sharing, and managing incidents with upstream and downstream partners is a critical component of supply chain agility.
November 13, 2020
Does this scenario sound familiar? The supplier manager at a large pharmaceutical company receives a notification about a critical active pharmaceutical ingredient (API) shortage that is impacting a supplier halfway around the world—and it instantly sends the team scrambling to find an alternative supplier so the product can be manufactured and delivered on time, in full (OTIF).
Achieving supply chain agility requires capabilities that will help you ensure much tighter, more transparent, and holistic relationships with suppliers, as well as structured collaboration that has not been consistently achieved in the pharmaceutical industry. - IDC
The team races to connect with the API supplier, the contract manufacturer, alternative suppliers, and downstream supply chain stakeholders to collaborate and organize around schedule changes, production delays, the material shortage, and other issues impacting the on-time delivery of the order. The messy and time-consuming process involves many phone calls, emails, and faxes, and it requires stakeholders to access multiple, siloed systems to manage through the incident.
These types of situations would go much more quickly and smoothly if all stakeholders adopted some of the key capabilities—like a single platform for incident management and data sharing—that help ensure higher levels of supply chain agility and responsiveness, according to a new report from IDC.
IDC and TraceLink surveyed more than 500 pharma supply chain companies and found that supply chain agility and proactive incident management is severely lacking. More than 43% of respondents report that they lack the agility needed to efficiently respond to and manage major supply chain disruptions. The chart below shows the top initiatives that pharma supply chain companies are doing or plan to do to become more agile.
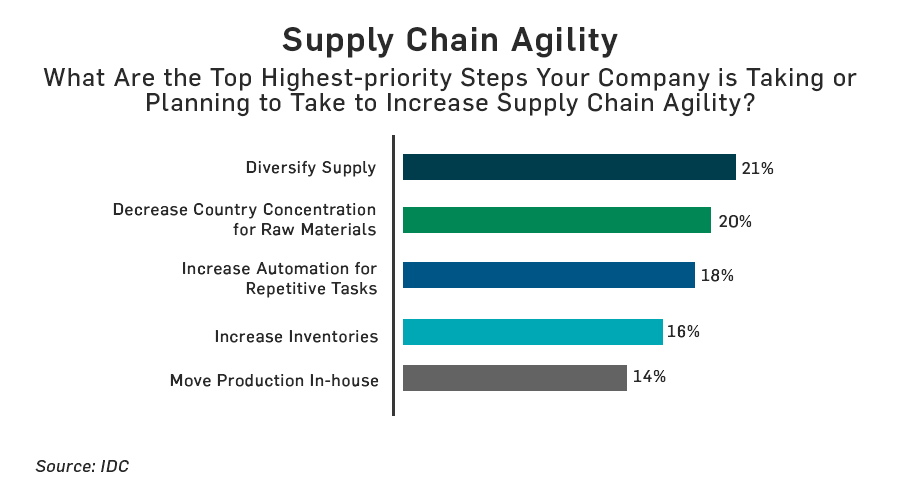
Top capabilities for achieving pharma supply chain agility
Achieving supply chain agility requires capabilities that will help you ensure much tighter, more transparent, and holistic relationships with suppliers, as well as structured collaboration that has not been consistently achieved in the pharmaceutical industry. Here’s a list of some of the key tools and capabilities IDC says you’ll need to develop to be more proactive and achieve greater agility in your end-to-end supply chain.
- A multi-enterprise platform for collaboration and data sharing
A key component of supply chain agility is enabling various teams to work together to resolve issues that arise—and not just teams within your own organization, according to IDC’s Simon Ellis, who authored the whitepaper. You need the ability to collaborate and share data seamlessly with all upstream and downstream partners on a single digital platform that enables faster incident management and issue resolution, as well as the discovery of new business insights that can be leveraged to boost agility and efficiency even further.
- End-to-end supply chain visibility
A supply chain that has both actionable visibility and contextually accurate supply chain agility will be resilient to disruption more often than those that do not, Ellis points out. Tools that can help you increase supply chain visibility include multi-enterprise business network, analytics, and IoT technologies. But technology alone will not enable agility. You also need to codify business practices and integrate them into a faster-reacting business model to support supply chain agility.
- Responsiveness
It’s not enough to gain visibility into the occurrence and potential impact of supply chain disruptions, you also need the ability to take swift action, IDC advises. Companies require capabilities that enable them to quickly assess and responsively manage the impact of disruptions, and then take the learnings from that experience and incorporate them into operations in a reliable and compliant way. IDC says this will help increase efficiently by ensuring that the problem does not resurface or simply move to another node of the end-to-end supply chain.
Want to learn more about the capabilities you need to achieve supply chain agility? Download the IDC white paper “Supply Chain Agility in the Pharmaceutical Industry” today.