DSCSA for Manufacturers
Thank you for contacting us; we’ll be in touch shortly.

Case Study: Noden Pharma | The Cost of Non-Compliance
See how global pharmaceuticals company Noden Pharma avoided the financial and operational risks of DSCSA noncompliance.

Saleable Returns: VRS Capabilities and Decision Criteria for Manufacturers
Learn the criteria for choosing a VRS as the foundation of a saleable returns solution, including network governance, scalability, and…

DSCSA Verification: VRS Essentials for Manufacturers and Wholesalers
Before selecting a Verification Router Service to meet 2019 DSCSA requirements, see what wholesales and manufacturers need to ask.

Case Study: Sharp Packaging Services | EPCIS Connection Changes Post Go-Live
See how Sharp Packaging Services overcame EPCIS change management challenges in the pharma supply chain with TraceLink's help.

Solving Saleable Returns: Critical Steps Toward Meeting the Deadline
In this webinar series, TraceLink experts will guide you through the maze of saleable returns and product verification:

Aggregation: Why is it Such a Hot Topic?
While aggregation isn’t a regulatory requirement in the U.S., understand how the expectations of your wholesale distributor customers may impact your…

Preparing for DSCSA Saleable Returns Verification
Understand the impact of the upcoming DSCSA saleable returns verification requirement for both pharma companies and wholesale distributors.
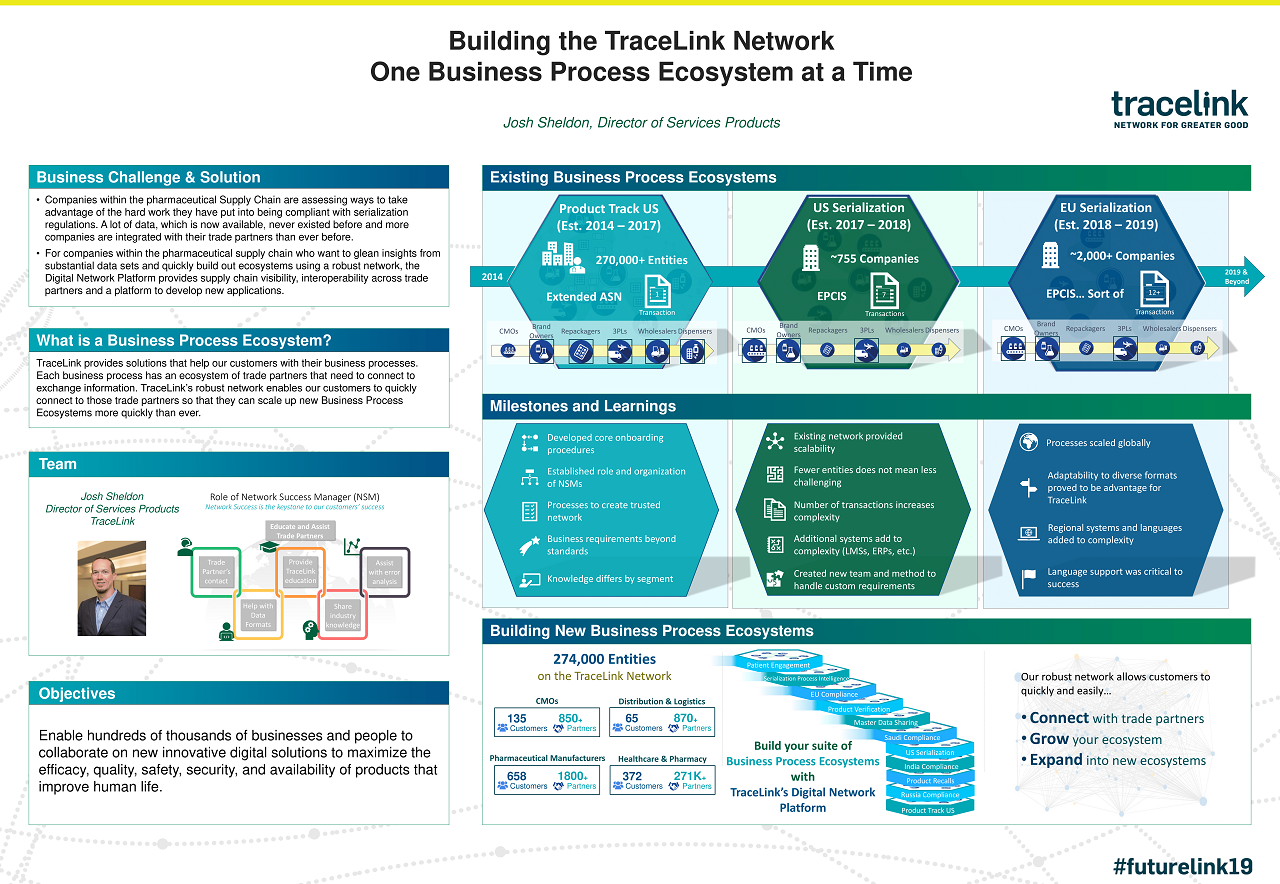
Case Study: TraceLink | Building the TraceLink Network One Business Process Ecosystem at a Time
See how TraceLink's powerful digital supply network enables customers to quickly connect to trade partners for scaling up business process ecosystems.

Transforming the Health System Pharmacy Supply Chain Through DSCSA
Watch this on-demand webinar to learn how health system pharmacies are driving new value from compliance.

Solving the Challenge of Trade Partner Requirements
Find out what happened when a wholesaler turned to TraceLink after getting an unexpected request.

Saleable Returns: DSCSA and Trade Partner Requirements Explained
View this on-demand webinar on saleable returns, DSCSA requirements and the role of master data.

Case Study: TraceLink | FDA Pilot - Product Traceability Under 2023 DSCSA Regulations - A Business Process-Led Design for a Blockchain Network
TraceLink's breakthrough blockchain solution, Trace Histories, can help pharma customers comply with US DSCSA regulations that go into effect in 2023.

