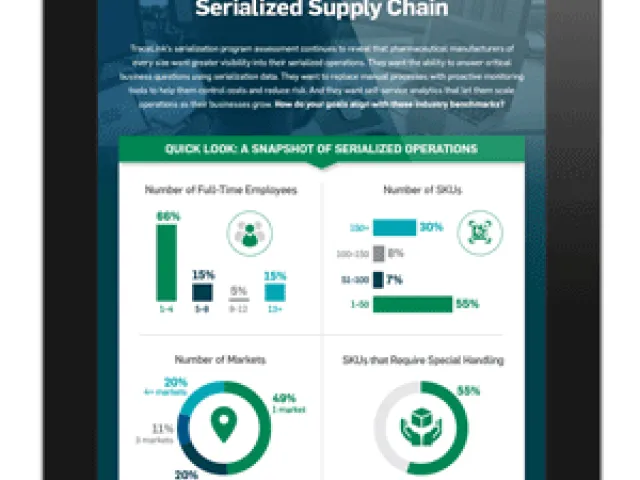
Table of contents
China’s amended Drug Administration Law introduces new systems, new data and reporting requirements, new serialization options—and new urgency for companies doing business in China. The National Medical Products Administration is acting very quickly to put these new requirements in place, beginning with an intermediate deadline on December 31, 2020 and with additional intermediate deadlines expected in the lead up to full traceability in 2022.
In this interview, TraceLink’s Director of Industry Marketing and Community, Allan Bowyer, discusses what companies need to be doing now to ensure long-term success in meeting China’s evolving regulatory and business requirements.
Why do changes to the Chinese regulations mean that companies will have to change the way they generate reports for the China market?
Allan Bowyer: Chinese regulations for medicines are indeed changing. In fact, at the end of 2019, the Drug Administration Law—or DAL—was amended. You may know there was a traceability regulation in China between 2013 and 2015 that required serialization and some reporting. The new law brings in further options for that serialization, and it greatly expands the scope of reporting and the data that's required to do that reporting.
To support the national government's vision for this, and with the growing empowerment of the provinces in China as well, they've designed a three-tier reporting system, so even the reporting system is changing. Manufacturers, importers, and others across the supply chain will be looking at changes to not only serialization practices, but the types and volumes of reports that manufacturers and others generate. The sheer amount of information they will need to send will increase.
What's the biggest difference between the old and new reporting requirements under the revised laws in China?
AB: While there are a number of differences between the old set of reports and the new set of reports, at a high level, the number of reports in the old regulations were quite minimal in comparison to the number of reports and the data that goes into them for the new regime.
In looking at the new China reporting requirements, TraceLink did an analysis, looking at a manufacturer who is importing products into China. We took a look at how “fit for purpose” the data would be using the reports for the previous regime. In that scenario alone, we found that there were 35 fields that were not in the old reports that are in the new reports. Beyond the increased data requirements, businesses need to ask, "Where are we going to source that data from, and how can we report that data in a consistent, seamless manner in the future?"
Why shouldn't manufacturers accept the current way of doing things as they look forward to the new China regulations?
AB: The new Drug Administration Law really does cover many facets. It's an entire revamp of the supply chain in China in terms of traceability and quality. Companies must not assume that compliance with the old regime will bring them into compliance with the new regime. The old reports simply do not meet the requirements of the new traceability systems. From our own analysis, we've seen that reports have changed, the data has changed, and the serialization options have changed. With the new system, there's likely to be new schema and other requirements as well at a technical level.
I would encourage local affiliates and their global counterparts to act quickly, to analyze their business requirements, and to work with their affiliates to understand what they will be accountable for in terms of events, in terms of data, and in terms of sequencing their operations in order to ensure that they are prepared for this new traceability system and traceability reports.
What are the key capabilities companies must look for when they are choosing a traceability solution partner in China?
AB: As part of your China requirements, you will need to report to the NMPA’s Harmonization Service Platform, also referred to as the Collaborative Service Platform. More often than not, that means partnering with a third-party traceability provider.
It's important to understand that processes will change, operations will change, and systems will change under the new system. A very important consideration is that the new law provides options for a product coding strategy. With the old regime, you had to use the China eCodes. Now, the law is giving an option to use a mix-and-match strategy with those codes and GS1 coding. The global GS1 standard, of course, offers a great amount of flexibility, power, and consistency, but what we've seen is that it's not just about the law, it's about the entire distribution ecosystem.
TraceLink supports both the EDMC coding and GS1 because we've worked with multiple markets, including China. You don't want to box yourself in early with a solution provider who can only support one of those codes. We're encouraging our customers to work with their trade partners to ensure that any coding scheme that they do adopt can be changed to ensure that—in the future—all your products can be read and traced once they leave your control.
How is TraceLink helping companies prepare for the new China reporting requirements?
AB: If you are selling products into China or you're a manufacturer importing products into China you need to map out your business operations—the financial ownership and the physical ownership of your goods—very carefully. That includes the licensure as well, because all of these are very important in defining your reporting requirements, and hence your data requirements. TraceLink can help you with that mapping, and we can help you discover requirements you may not have known that you had.
We are working right now with customers to understand, because of the expanded scope of the Drug Administration Law, how their business operations will change, and consequently, how their reporting obligations will change. We believe that by bringing heads together, by bringing industry leaders together, we can come up with the best answers and navigate the ambiguity that is quite present with China.
We have a customer community that meets and discusses the interpretation of new requirements, how companies are approaching those requirements in terms of their operations, looking at the pros and cons of any one approach. Essentially, we have a brain trust of industry expertise that we can call on to help prepare others for their traceability journey in China.
TraceLink: A proven partner in China compliance solutions
Staying up to date on the complexity of China’s regulatory requirements is a significant challenge—even for companies with experience in other global markets. Since 2013, TraceLink has been helping companies meet the original China serialization and traceability regulations with its China compliance solutions—and continues to provide unmatched industry expertise for its customers through the TraceLink Community of operational leaders, regulatory experts, and technical innovators.