Table of contents
The lot-level requirements of DSCSA (Drug Supply Chain Security Act) have been in effect for hospitals and pharmacies since July 2015, as the law’s first step in keeping illegitimate drugs away from patients. But for many healthcare organizations, managing compliance-related tasks has had a negative effect on other aspects of their business.
Hospitals and pharmacies shared the details of their challenges in a live poll during our July webinar on the burden of DSCSA compliance work, attended by 110 healthcare companies across the U.S.
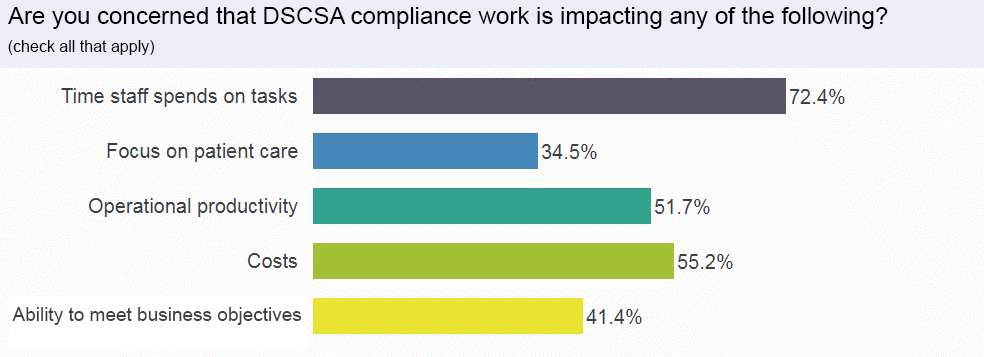
Nearly 3 of 4 respondents are concerned about the time pharmacy staff is spending on DSCSA tasks.
The amount of time healthcare organizations are spending on DSCSA work can vary, of course, depending on the size of the pharmacy operation and the volume of their inventory and receiving. The majority of poll respondents are spending up to 5 extra hours a week on compliance tasks, with medical centers and health systems spending as much as 10 additional hours per week.
While time spent on DSCSA work was named the greatest concern, the cost of compliance and a loss of productivity were worrisome for more than half of all respondents.
Cost can be a factor beyond just current lot-level compliance. Looking ahead, organizations using a compliance solution that hasn’t been developed for serialization may feel the financial impact within the next 2 years, when they’ll need to acquire an add-on, or a second solution, to meet serialization requirements.
If your hospital or pharmacy is ready for serialization now—serialized product will be at your door by November, if not already—you can take advantage of the ways serialized data can improve your business. Webinar attendees were asked where their businesses could realize value from serialization.
100% of respondents said that recall notifications would be a benefit of serialization data.
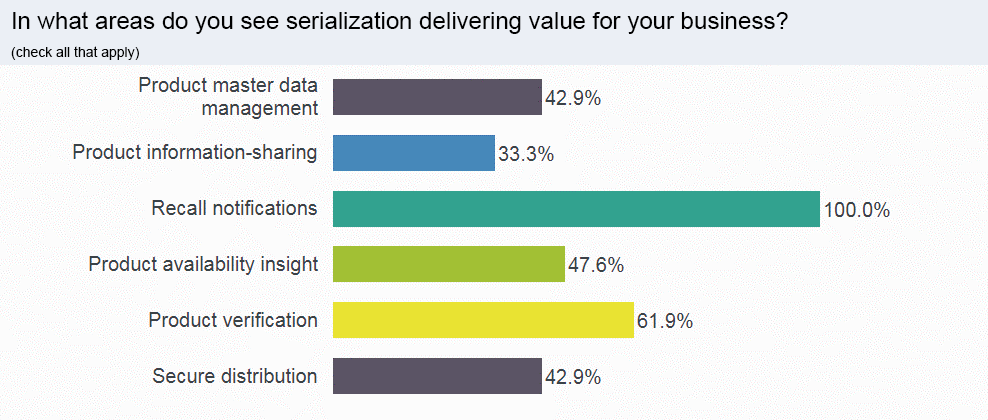
That unanimous response underscores how effectively accessible, serialized data can streamline and improve the recall process—and the inherent challenges in how recalls are currently managed.


