blue
Product overview
Verify product with the speed and scale required by compliance mandates
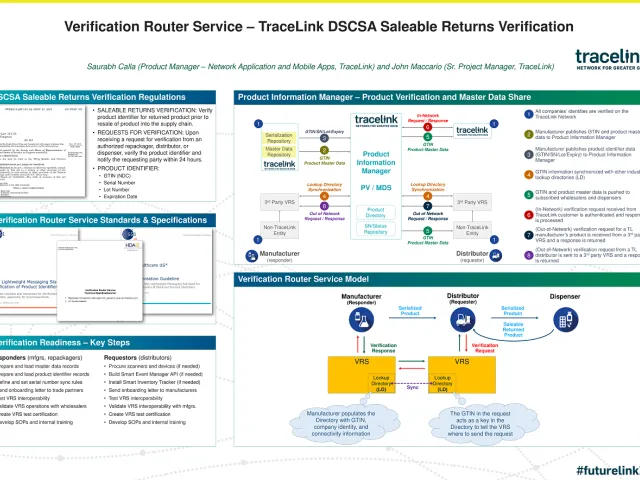
Verify serialized product identifiers and their status across both direct and indirect supply network relationships in support of new business and regulatory requirements. With the secure publication of serialized product information into a network-facing repository, network entities purchasing, distributing, or otherwise handling a manufacturer's products can interrogate their serialized product identity as needed with the speed and scale demanded by today's supply chain.
Product Information Manager - Product Verification provides:
Industry challenges
New regulatory mandates create a new product verification requirements
Manufacturers, repackagers, wholesalers, and pharmacy dispensers face a range of new product verification requirements under the U.S. Drug Supply Chain Security Act (DSCSA). Verification requests must be answered quickly to ensure the timely movement of product and to avoid noncompliance. Challenges include:
An influx of product verification requests
There are many scenarios in which network entities could be required to verify product identity, and the number of verification requests received on a daily basis is highly variable. Furthermore, requests could come in from trade partners or federal agencies like the FDA.
An influx of product verification requests
Demand for sub-second product verification
Manual verification is not a viable approach under the new DSCSA requirements. The U.S. pharmaceutical industry has mandated product verifications must be completed in less than one second.
Demand for sub-second product verification
Recalled or expired products risk supply chain safety
The safety and efficiency of the product supply chain is always under threat from recalled or expired products moving unnoticed through product supply operations. Identification and removal of such products improves patient safety.
Recalled or expired products risk supply chain safety
Capabilities
Secure the supply chain and ensure compliance with high-speed product verification
Product Information Manager - Product Verification provides a secure, efficient solution for verifying serialized product identifier data in a network-facing application.