Table of contents
More than 125 companies have selected its Verification Router Service (VRS) to meet the saleable returns verification requirement by the November deadline
TraceLink Inc., the world’s largest integrated digital supply network, today announced its continued momentum in helping companies across the supply chain comply with Drug Supply Chain Security Act (DSCSA) requirements, leading the industry in its commitment to an open, interoperable, standards-based approach.
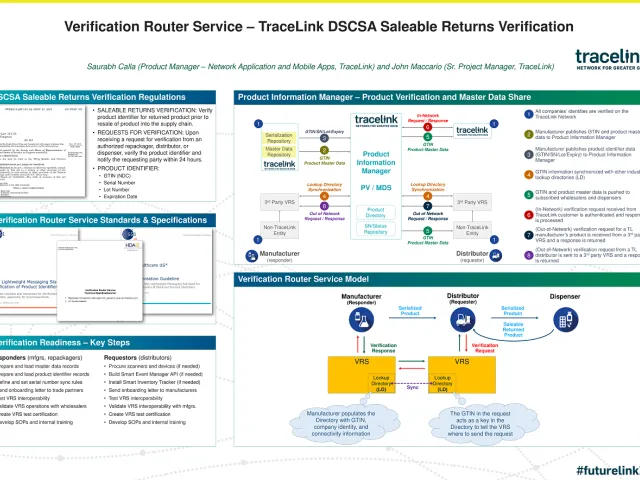
Over 125 pharmaceutical manufacturers and wholesale distributors have selected TraceLink's saleable returns verification solution, which includes a verification router service (VRS) and master data sharing capabilities built on its Product Information Manager network application. For pharmaceutical manufacturers, TraceLink's saleable returns verification solution addresses the challenge of distributing product master data, including package level specifications to direct and indirect trading partners so that they can receive aggregated, serialized product, while the VRS enables the ability to respond to wholesaler verification requests. For wholesale distributors, the solution is used to collect product and package level specifications from their suppliers, using mobile scanning for return receiving and VRS to look up the location of a manufacturer’s VRS and to submit verification requests.
To demonstrate its preparedness, TraceLink recently completed a series of successful round-trip verification tests together with multiple wholesalers, manufacturers, and other solution providers across a variety of testing scenarios and data configurations. These tests further reinforced the benefits of following and implementing open, interoperable standards to support saleable returns verification across a wide variety of technical architectures and solution providers. While scalability and performance were not a focus of this testing, TraceLink noted that roundtrip verification request/response times using TraceLink’s Product Information Manager were completed well within the "sub-second” response time.
“Our recent interoperability testing confirmed the benefits of TraceLink’s proven network approach and we will continue to invest and innovate in ways that will help our customers realize the value of their compliance investments by solving new and long-standing challenges through the end of DSCSA and beyond,” said Shabbir Dahod, president and CEO, TraceLink. “Since the early days of DSCSA, the success of our customers, and of the industry has been TraceLink’s priority and is what drives our commitment to open standards, transparency, and interoperability, all of which support an efficient and collaborative pharmaceutical supply chain.”
As the industry continues its journey towards full unit-level traceability across the supply chain, it will be required to establish fundamental standards in network connectivity, data exchange, and shared business processes, as well as further define a governance of standards for network adoption. TraceLink has demonstrated its commitment towards this goal by recently joining the new governance organization for DSCSA interoperability proposed by the Pharmaceutical Distribution Security Alliance (PDSA).
This week, TraceLink is at the HDA Traceability Conference in Washington, D.C. Visit TraceLink booth 23 to discuss saleable returns, and learn more about TraceLink’s proven network approach to DSCSA compliance.