blue
Product overview
Comply with Brazil’s serialization and reporting requirements
TraceLink Brazil Pharmaceuticals enables multinational and Brazilian domestic pharmaceutical manufacturers to meet ANVISA requirements for product serialization and compliance reporting to the centralized SNCM (Systema Nacional de Controle de Medicamentos) system.
With Brazil Pharmaceuticals, you can:
Industry challenges
New compliance requirements demand new compliance solutions
Today’s marketing authorization holders and pharmaceutical manufacturers face immense challenges in managing regulatory compliance requirements emerging in the Brazilian market. These challenges include:
Complex serialization and reporting requirements
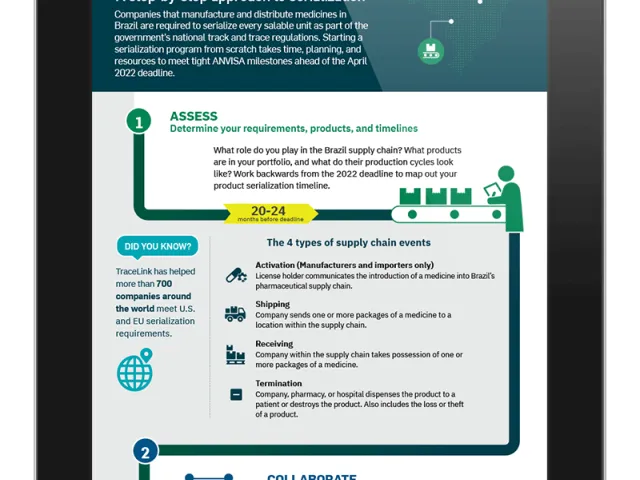
Brazil’s complex end-to-end traceability regulations affect manufacturers, importers, distributors, and dispensers alike, and each segment has unique reporting requirements.
Complex serialization and reporting requirements
Siloed compliance processes and reporting
The central government system does not connect trade partners to work on multienterprise processes, such as tracking and resolving shipping and receiving errors.
Siloed compliance processes and reporting
Incorrect compliance data
Reporting and data errors can lead to product delays if they aren’t resolved quickly between trading partners.
Incorrect compliance data
Capabilities
TraceLink Brazil Pharmaceuticals
This solution provides the full compliance data management capabilities companies need to build a secure and flexible Brazil serialization and traceability compliance infrastructure. You can: